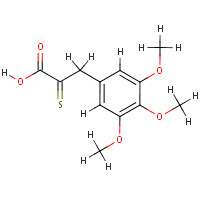
| Physical Properties | | | |
| Molecular Weight: 269.297 g/mol | logS: -3.41197 | SlogP: 0.37467 | Reactive groups: 0 |
| | | | |
| Topological Properties | | | |
| Globularity: 0.102269 | Sterimol/B1: 2.4877 | Sterimol/B2: 3.39543 | Sterimol/B3: 3.56495 |
| Sterimol/B4: 8.88636 | Sterimol/L: 12.0409 | | | |
| | | | |
| Surface and Volume Properties | | | |
| Accessible surface: 479.831 | Positive charged surface: 320.476 | Negative charged surface: 159.355 | Volume: 241.875 |
| Hydrophobic surface: 324.045 | Hydrophilic surface: 155.786 | | |
| | | | |
| Pharmacophoric Properties | | | |
| Hydrogen bond donors: 0 | Hydrogen bond acceptors: 4 | Acid groups: 2 | Basic groups: 0 |
| Chiral centers: 0 | | | |
| | | | |
| Drug- and Lead-like Properties | | | |
| Lipinski's drug-like rule: 1 | Violations of Lipinski's rule: 0 | Oprea's lead like rule: 1 | |