|
 |
|
 |
Search | Help | MolPaint | Roadmap | Credits | Feedback |
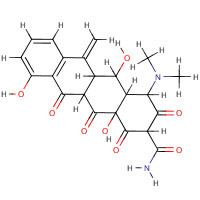
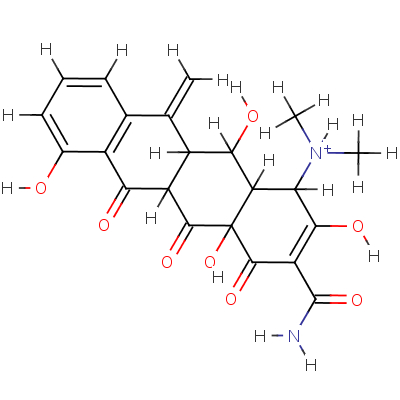
| SMILES: | OC12C(C([NH+](C)C)C(O)=C(C(=O)N)C1=O)C(O)C1C(C2=O)C(=O)c2c(c ccc2O)C1=C |
| InChI: | InChI=1/C22H22N2O8/c1-7-8-5-4-6-9(25)11(8)16(26)12-10(7)17(27)14-15(24(2)3)18(28)13(21(23)31)20(30)22(14,32)19(12)29/h4-6,10,12,14-15,17,25,27-28,32H,1H2,2-3H3,(H2,23,31)/p+1/t10-,12-,14-,15-,17-,22+/m1/s1 |
 |
|
| download 2D Mol File
| download 3D Mol File
Potential Energy Epot(MMFF94)=60.5502 kcal/mol |
MOE's Descriptors
| Physical Properties | ||||||
| Molecular Weight: 443.432 g/mol | logS: -2.75565 | SlogP: -2.4818 | Reactive groups: 1 | |||
| Topological Properties | ||||||
| Globularity: 0.289974 | Sterimol/B1: 2.43004 | Sterimol/B2: 3.5964 | Sterimol/B3: 7.3862 | |||
| Sterimol/B4: 8.15351 | Sterimol/L: 13.5427 | |||||
| Surface and Volume Properties | ||||||
| Accessible surface: 606.167 | Positive charged surface: 393.162 | Negative charged surface: 213.005 | Volume: 380.625 | |||
| Hydrophobic surface: 284.266 | Hydrophilic surface: 321.901 | |||||
| Pharmacophoric Properties | ||||||
| Hydrogen bond donors: 6 | Hydrogen bond acceptors: 8 | Acid groups: 0 | Basic groups: 1 | |||
| Chiral centers: 6 | ||||||
| Drug- and Lead-like Properties | ||||||
| Lipinski's drug-like rule: 1 | Violations of Lipinski's rule: 1 | Oprea's lead like rule: 0 | ||||
|